Magnetic resonance imaging can now reveal the structure and function of the brain in extraordinary detail. But how can researchers detect the subtle changes hidden among thousands of data points that precede the onset of Alzheimer’s or other neurodegenerative diseases? Artificial intelligence is entering medicine in ways that would have seemed like science fiction just a few years ago - and Milan Němý of CIIRC ČVUT is using these tools to search for answers. In his research, he combines advanced imaging methods with AI techniques to uncover patterns in data that are invisible to the human eye. On the occasion of Brain Awareness Week, we bring you an interview about how brain scans can be “read” in new ways - and what this could mean for the future of early diagnosis.
At the end of 2025, Milan Němý was selected by an international committee for a new faculty position within the European Centre of Excellence CLARA Centre, which focuses on connecting artificial intelligence, bioinformatics, and brain research. This appointment provides additional institutional support for his ambitious goal: to understand better how the brain changes in the earliest stages of disease - and how these changes can be detected early and reliably.
You work at the intersection of AI, brain imaging, and neuroscience. How would you describe your research to someone outside the scientific community? And what real-world problems are you ultimately trying to solve?
The focus of my research is understanding how the brain changes long before diseases like Alzheimer’s develop visible symptoms. In our work, we mainly take advantage of modern, advanced magnetic resonance imaging (MRI) and artificial intelligence tools to look for very subtle alterations in brain structure and connectivity, signals that are often invisible to the naked eye.
In simple terms, we try to teach computers to “read” the brain MRI scans more precisely than humans can. The ultimate goal is to detect vulnerability early, understand which brain systems are affected, and predict how the disease might progress, both in time and across different parts of the brain. If we can identify risk earlier and more accurately, we move closer to prevention and personalized management rather than crisis-driven care.
Many brain diseases, including Alzheimer’s disease and brain cancer, are still diagnosed relatively late. What is the core challenge your research is trying to address, and how does AI help overcome it? Why is MRI such a powerful tool in this context?
The core challenge is that brain diseases develop gradually and silently. By the time symptoms become obvious, significant and often irreversible damage has already occurred.
In general, MRI is a powerful tool as it allows us to look inside the living brain non-invasively and quantify its structure in detail. However, we are already past the phase of thinking of MRI as just a non-invasive way to visualize the anatomy of the human body. In the context of our research, we use MRI as a powerful physical “analyzer” that captures microscopic tissue properties and spatial information about brain networks. This helps us detect early disease signals that are subtle and distributed as they do not appear as simple, easily visible lesions.
To analyze large datasets of MRI images across multiple sequences (together with other patient data), AI is now essential. Our machine learning models can identify combinations of brain features and trajectories that would be impossible to recognize manually. Instead of asking, “Is this brain already damaged?”, we can begin asking, “Is this brain at risk, and how is it changing over time?”
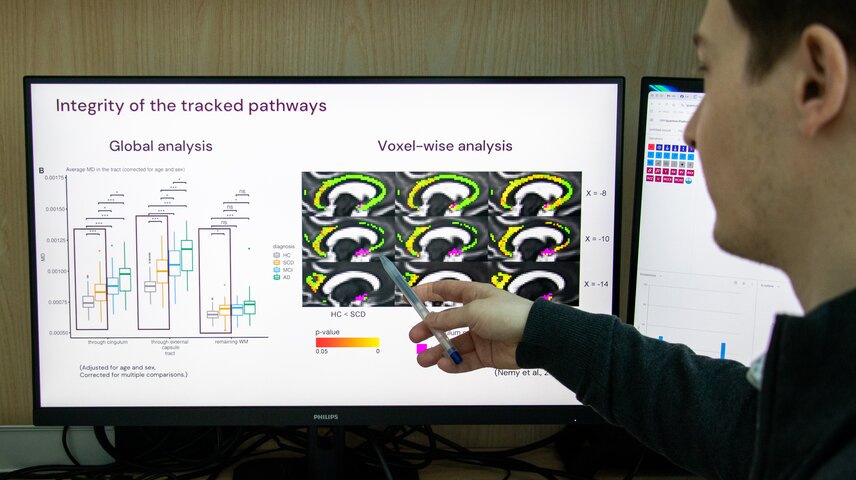
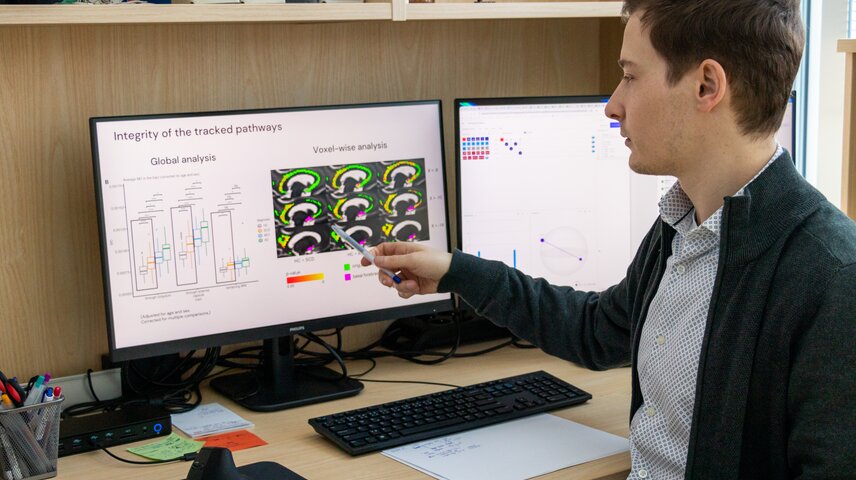
A key part of your work focuses on subtle changes in the brain, such as those affecting the cholinergic system in Alzheimer’s disease. What have we been missing until now, and what new insights are these methods allowing you to uncover?
Traditionally, Alzheimer’s research has focused on hallmark markers such as amyloid, tau, or hippocampal atrophy. While these are important, they do not fully explain early cognitive vulnerability.
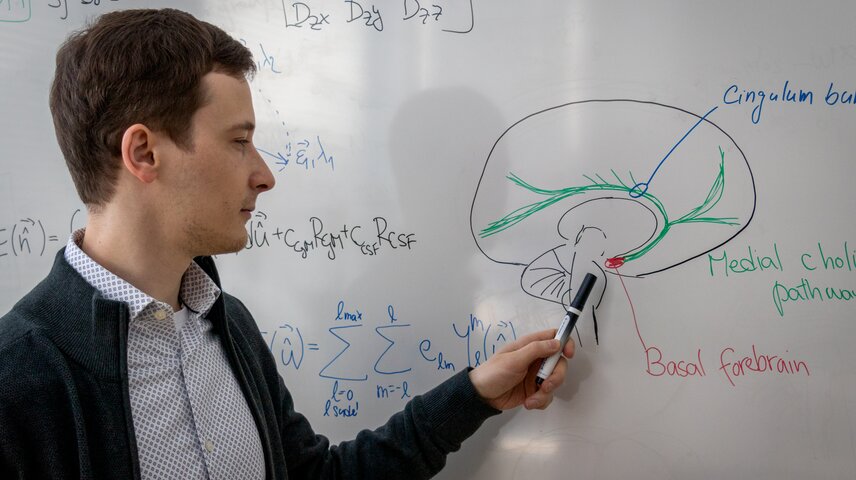
What has been largely overlooked is the degeneration of specific brain networks that regulate attention, memory modulation, and cognitive flexibility, such as the cholinergic system. When it is disrupted, many other brain functions can become affected as well. However, these pathways are spatially complex and difficult to quantify using conventional imaging methods. For a long time, the field has struggled with precise identification of this system in patients’ brains and with having methods sensitive enough to capture subtle but important changes.
In our work, we developed advanced segmentation techniques for the cholinergic system. By combining them with diffusion MRI and AI-driven analysis, we can now quantify the integrity of these networks in vivo. This was not possible before. It opens the door to a whole new area of Alzheimer’s disease research. With this method in our hands, we can move from investigating the consequences of the disease to investigating its origins. For example, it allows us to study younger individuals at an age when pathophysiological changes may begin, but where traditional methods were not sensitive enough.
You were recently appointed to a new CLARA tenure-track faculty position in Bioinformatics and AI. What attracted you to CLARA, and how does this position support your scientific independence and long-term research vision?
What personally appeals to me about CLARA is that it brings together experts from many different fields, such as artificial intelligence, high-performance computing, quantum technologies, and neuroscience, under one framework. In my opinion, this synergy is essential for tackling complex problems like neurodegeneration, which cannot be solved within a single discipline. As self-evident as this may sound, it is still not very common in science. That is why I truly appreciate the initiative CLARA is pursuing.
On a practical level, the tenure-track position provides me with the scientific independence to develop my own research direction, while also being part of a collaborative ecosystem with strong computational and technological infrastructure. I believe this combination can support a long-term, high-quality line of research, and help build a team with continuity over time.
Looking ahead, what are your main research goals within CLARA over the next few years? Are there new directions, collaborations, or projects you are particularly excited about?
Over the next few years, my main goal within CLARA is to build a strong, interdisciplinary research team that brings together neuroscientists, AI specialists, radiologists, and neurologists. With CLARA’s infrastructure and support, I aim to develop a group that is scientifically independent and sustainable in the long term.
Scientifically, one major priority is further validation of our analytical framework for assessing the cholinergic system. We are now extending this work through PET imaging, longitudinal cohort analyses, and involvement in drug trials. We already have international collaborators for each of these directions, and we are excited to extend these partnerships to test our methods in diverse populations and real clinical settings.
Another key direction is addressing co-pathologies, particularly cerebrovascular disease, which plays a critical role in cognitive decline but remains difficult to quantify comprehensively. Currently, there is no robust, standardized tool for evaluating the spatial distribution and characteristics of cerebrovascular lesions at scale. We are developing a novel, high-tech analytical framework that combines advanced imaging with machine learning to characterize vascular brain damage in a more systematic and biologically meaningful way. This would not be possible without strong technical expertise in AI and imaging. Our goal is to make this tool available to the research community, and potentially, in the future, to clinical practice.
Most of these efforts are carried out through international collaborations. These partnerships bring together complementary expertise and, importantly, access to new patient datasets that are essential for validation and generalization of AI-based models.
Neurodegenerative diseases are one of the major challenges of aging societies. From your perspective, what role can AI-driven brain research play in changing how these diseases are diagnosed or managed in the future?
From my perspective, I really think AI-driven brain research can make a difference. Over the last few years, I’ve seen AI-supported work clearly accelerate research and reveal patterns and knowledge we simply didn’t have access to before.
In the best-case scenario, especially in the context of basic science, AI-supported neuroscience could help uncover underlying principles that genuinely revolutionize our understanding of these diseases, to the point where prevention becomes possible. We have seen similar paradigm shifts in other fields, although of course it’s hard to predict if and when that will happen in neurodegeneration.
What I think is more likely in the near future is a very practical transformation: with AI-driven analysis of MRI and other biomarkers, we will be able to identify individuals at risk much earlier than today. That can enable targeted monitoring, personalized prevention strategies, and better timing of therapeutic interventions. It can also improve clinical trial design for new drugs by selecting the right participants and using more sensitive outcome measures. Overall, this is a step toward precision neurology, where decisions are guided by the biology of an individual brain rather than broad diagnostic categories.
Latest publications
- Rennie, A., Nemy, M., Jerele, C., Rodríguez-Baz, I., Montal, V., Bejanin, A., … & Ferreira, D. (2025). Regional associations between cerebrovascular disease and cholinergic white matter pathways in the Lewy body continuum. npj Parkinson’s Disease, 11(1), 250.
- Pažout, J., Němý, M., Mikeš, J., Jirman, J., Kubr, J., Niebauerová, E., … & VentConnect Study group. (2025). SmartAlert: Machine Learning-Based Patient-Ventilator Asynchrony Detection System in Intensive Care Units. Computer Methods and Programs in Biomedicine, 108927.
- Jerele, C., Tzortzakakis, A., Nemy, M., Rennie, A., Arranz, J., Montal, V., … & Ferreira, D. (2025). Cerebrovascular co-pathology and cholinergic white matter pathways along the Lewy body continuum. Brain Communications.
- Sedlák, V., Němý, M., Májovský, M., Bubeníková, A., Nordin, L. E., Moravec, T., … & Netuka, D. (2025). IDH Status in Brain Gliomas Can Be Predicted by the Spherical Mean MRI Technique. American Journal of Neuroradiology.

Ing. Milan Němý, Ph.D.
CIIRC CTU
Milan Němý leads a neuroscience research group at the Czech Institute of Informatics, Robotics and Cybernetics at CTU in Prague (CIIRC CTU), within the Department of Cognitive Systems and Neuroscience (COGSYS). He is also a researcher at Karolinska Institutet in Sweden, one of the world’s leading medical universities and the institution responsible for awarding the Nobel Prize in Physiology or Medicine.
His work focuses on neurodegenerative diseases, particularly Alzheimer’s disease, and on the use of advanced MRI methods combined with artificial intelligence. His research contributes to a better understanding of early brain changes and to the development of more sensitive biomarkers for early diagnosis.
He has received several prestigious awards, including the Jan Bureš Award 2023 for the best scientific article in neuroscience, the GAČR Junior Star grant in 2024 to establish his own research team, and the Junior Faculty Award at the AD/PD™ 2025 international conference.
At CIIRC CTU, he holds a tenure-track position co-funded by the European Center of Excellence CLARA, where he develops research at the intersection of artificial intelligence, bioinformatics, and neuroscience to improve early diagnosis and understanding of brain diseases.
Milan Němý's page at CIIRC CTU